Advances in Peptide Synthesis Technologies and Their Applications in Modern Biotechnology
Advances in Peptide Synthesis Technologies and Their Applications in Modern Biotechnology
Introduction
Peptides are biologically active molecules composed of amino acids linked through peptide bonds. They play essential roles in cell signaling, immune regulation, and neurotransmission, making them critical components in both physiological processes and biomedical research.
The ability to synthesize peptides with high precision has significantly advanced our understanding of structure–function relationships, while also enabling the development of therapeutic peptides, biomaterials, and diagnostic tools. Over the past century, peptide synthesis technologies have evolved from classical chemical approaches to highly sophisticated and automated systems, with solid-phase peptide synthesis (SPPS) emerging as the dominant methodology.
Evolution of Peptide Synthesis Technologies
Liquid-Phase Peptide Synthesis (LPPS)
Liquid-phase peptide synthesis represents the earliest approach to peptide production, developed in the early 20th century. This method involves stepwise coupling or fragment condensation in solution, where amino acids are sequentially assembled to form peptide chains.
Historically, LPPS enabled landmark achievements such as the synthesis of biologically active peptides, demonstrating the feasibility of producing complex molecules chemically. However, each reaction step requires isolation and purification of intermediates, making the process labor-intensive and difficult to automate.
Despite these limitations, LPPS still offers advantages:
- Controlled reaction conditions
- Flexible protection strategies
- Suitability for short peptides (<10 amino acids)
- Scalability for industrial applications
Ongoing developments aim to improve LPPS through automation, high-throughput strategies, and environmentally sustainable processes.
Solid-Phase Peptide Synthesis (SPPS)
Introduced in the 1960s, solid-phase peptide synthesis revolutionized peptide chemistry by simplifying purification and enabling automation. In this approach, the growing peptide chain is anchored to an insoluble solid support, allowing sequential addition of amino acids.
After each reaction step, excess reagents and by-products are removed by simple washing, eliminating the need for complex purification procedures. This innovation significantly improved efficiency and reproducibility.
SPPS is now widely recognized as the standard method for peptide synthesis, supporting major advances in:
- Peptide-based therapeutics
- Vaccine development
- Proteomics and molecular biology
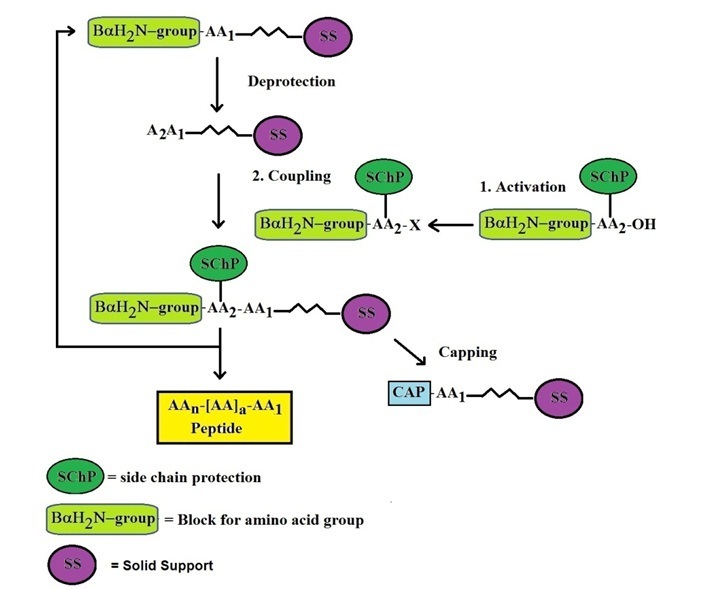 Principles of Solid-Phase Peptide Synthesis
Principles of Solid-Phase Peptide Synthesis
1. Solid Support Selection
The solid matrix plays a crucial role in synthesis efficiency. Ideal supports must:
- Be chemically stable
- Allow reagent diffusion (good swelling properties)
- Provide functional groups for peptide attachment
Common supports include:
- Polystyrene-based resins (robust but less flexible)
- PEG-modified resins (enhanced solubility and improved yield for long peptides)
2. Protecting Group Strategies
To prevent unwanted side reactions, amino acids are protected using specific chemical groups. The most widely used approach is the Fmoc/tBu strategy, which allows:
- Selective removal of protecting groups
- Compatibility with sensitive amino acid residues
- Precise control over peptide assembly
This concept of orthogonal protection ensures that different functional groups can be removed independently without affecting others.
3. Cyclic Synthesis Process
Peptide elongation occurs through repetitive cycles:
- Deprotection: Removal of the Fmoc group to expose the amino terminus
- Coupling: Addition of the next amino acid using activated reagents
- Washing: Removal of excess reagents and by-products
This cycle is repeated until the desired peptide sequence is fully assembled.
4. Cleavage and Purification
Once synthesis is complete:
- The peptide is cleaved from the solid support
- Protecting groups are removed simultaneously
- The crude product is purified, typically using HPLC
This results in a high-purity peptide suitable for biological applications.
Optimization Strategies in SPPS
Recent improvements focus on enhancing efficiency and product quality:
- Development of advanced protecting groups for sensitive peptides
- Use of high-performance coupling reagents to reduce side reactions
- Engineering of novel solid supports to minimize aggregation
- Optimization of synthesis conditions for difficult or long sequences
Advantages and Limitations of SPPS
Advantages
- Simplified purification process
- High reproducibility and automation
- High yield and purity for short peptides
- Compatibility with high-throughput synthesis
Limitations
- Reduced efficiency for long peptides (>50 amino acids)
- Aggregation issues in hydrophobic sequences
- High cost of reagents and materials
- Environmental concerns related to solvent use
Applications of Peptide Synthesis
1. Biomedicine
SPPS is widely used for producing peptide-based drugs targeting:
- Cancer
- Metabolic disorders
- Hormonal regulation
It also supports the development of peptide vaccines and therapeutic analogs.
 2. Protein Science
2. Protein Science
Synthetic peptides are essential tools for:
- Studying protein structure and function
- Investigating post-translational modifications
- Mapping binding sites and interactions
 3. Diagnostic Technologies
3. Diagnostic Technologies
Peptides are used to design:
- Highly sensitive biosensors (ELISA, SPR)
- Molecular probes for disease detection
4. Materials Science
Peptides contribute to innovative materials such as:
- Self-assembling hydrogels for tissue engineering
- Antimicrobial coatings for medical devices
5. Emerging Technologies Integration
SPPS is increasingly combined with advanced technologies:
- Enzymatic synthesis for greener processes
- Microfluidic systems for miniaturized high-throughput synthesis
- Computer-aided design (CAD) for optimized peptide sequences
- Automation and AI-driven synthesis platforms
Future Perspectives
Peptide synthesis technologies continue to evolve toward:
- Higher efficiency and scalability
- Reduced environmental impact
- Improved synthesis of complex and long peptides
Integration with artificial intelligence, automation, and green chemistry is expected to transform peptide production into a more precise, sustainable, and cost-effective process.
Conclusion
Advances in peptide synthesis, particularly the development of solid-phase methodologies, have significantly expanded the capabilities of modern biotechnology. SPPS stands as a cornerstone technology, enabling the rapid and reliable production of peptides for applications in medicine, research, and materials science.
Continued innovation will further enhance its role in addressing complex biological challenges and advancing next-generation therapeutic strategies.
Recent Posts
-
Advances in Peptide Synthesis Technologies and Their Applications in Modern Biotechnology
Advances in Peptide Synthesis Technologies and Their Applications in Modern Biotechnology …27th Mar 2026 -
Western Blot
Introduction Western blotting is a fundamental analytical technique widely used in molecular biology …27th Mar 2026 -
Ethidium Bromide as a Cooperative Effector of DNA Structure
Introduction DNA structure is not static. Under specific physicochemical conditions, DNA molecules c …25th Mar 2026