Electrophoresis Techniques in Molecular Biology
Electrophoresis is one of the most important analytical techniques used in molecular biology, biochemistry, and genetics. It allows researchers to separate and analyze charged biomolecules such as proteins and nucleic acids. Together with chromatography, electrophoresis forms the foundation of many laboratory workflows used for biomolecule characterization and purification.
This article explains the principles of electrophoresis, the factors affecting molecular migration, and the major electrophoretic techniques used in modern laboratories.
What Is Electrophoresis?
Electrophoresis is a laboratory method that separates electrically charged molecules based on their movement in an electric field.
When an electric current is applied across a conductive medium:
- Negatively charged molecules (anions) migrate toward the anode (+).
- Positively charged molecules (cations) migrate toward the cathode (−).
In biological systems, many macromolecules carry electrical charges:
- DNA and RNA possess negative charges due to their phosphate backbone.
- Proteins can carry positive or negative charges depending on their amino acid composition and the pH of the surrounding environment.
Because different molecules migrate at different speeds, electrophoresis enables their separation, identification, and characterization.
Basic Principles of Electrophoretic Separation
Electrophoretic migration depends on several physical and chemical parameters.
1. Molecular Charge
The net charge of a molecule strongly influences its migration. In proteins, this charge depends on the ionizable groups present in amino acids such as:
Negative charge contributors:
- Carboxyl groups of glutamic acid and aspartic acid
- Thiol groups of cysteine
- Hydroxyl groups of serine, threonine, and tyrosine
Positive charge contributors:
- Amino groups of lysine
- Imidazole group of histidine
- Guanidinium group of arginine
The pH of the medium determines whether these groups are protonated or deprotonated.
2. Isoelectric Point (pI)
The isoelectric point (pI) is the pH at which a molecule carries no net electrical charge.
- If pH > pI → molecule becomes negatively charged
- If pH < pI → molecule becomes positively charged
- If pH = pI → molecule does not migrate in the electric field
This concept is critical for techniques such as isoelectric focusing.
3. Electric Field Strength
Migration velocity is also proportional to the electric field strength applied. Higher voltage generally increases migration speed but may also cause heating of the system.
4. Molecular Size and Shape
Larger molecules experience more resistance when moving through a matrix such as gel, leading to slower migration.
This is why electrophoresis is commonly used to estimate:
- molecular weight of proteins
- size of DNA fragments
5. Properties of the Support Medium
The nature of the electrophoresis medium influences separation efficiency. Supports may include:
- liquid buffers
- paper
- cellulose acetate
- polymer gels such as polyacrylamide or agarose.
Porous gels act as molecular sieves, allowing smaller molecules to move faster than larger ones.
Major Electrophoresis Techniques Used in Laboratories
Different electrophoretic methods have been developed to analyze biomolecules under specific conditions.
1. Paper and Cellulose Acetate Electrophoresis

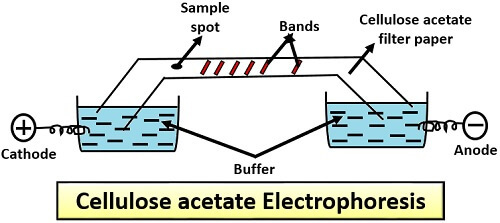
These are among the earliest electrophoretic methods.
Paper Electrophoresis
Paper electrophoresis uses filter paper as the support medium. Samples migrate along the surface of the paper under an electric field.
This method was historically used for separating:
- amino acids
- small charged molecules
However, it has largely been replaced by more advanced techniques.
Cellulose Acetate Electrophoresis
Cellulose acetate membranes provide a thin and simple support for electrophoresis.
Advantages include:
- low cost
- simple experimental setup
- rapid analysis
This technique is commonly used in clinical biochemistry, particularly for serum protein analysis. However, its resolution is relatively low compared to gel-based systems.
2. Polyacrylamide Gel Electrophoresis (PAGE)
Polyacrylamide gel electrophoresis is one of the most widely used techniques for analyzing proteins.
The polyacrylamide gel forms a three-dimensional mesh structure that acts as a molecular sieve. Molecules migrate through the gel depending on their size and charge.
Typical gel concentrations range from:
- 6%
- 8%
- 10%
- 12%
- 15%
Higher gel concentrations are used to separate smaller proteins.
PAGE is commonly used for:
- protein characterization
- enzyme analysis
- nucleic acid sequencing
- Western blot preparation
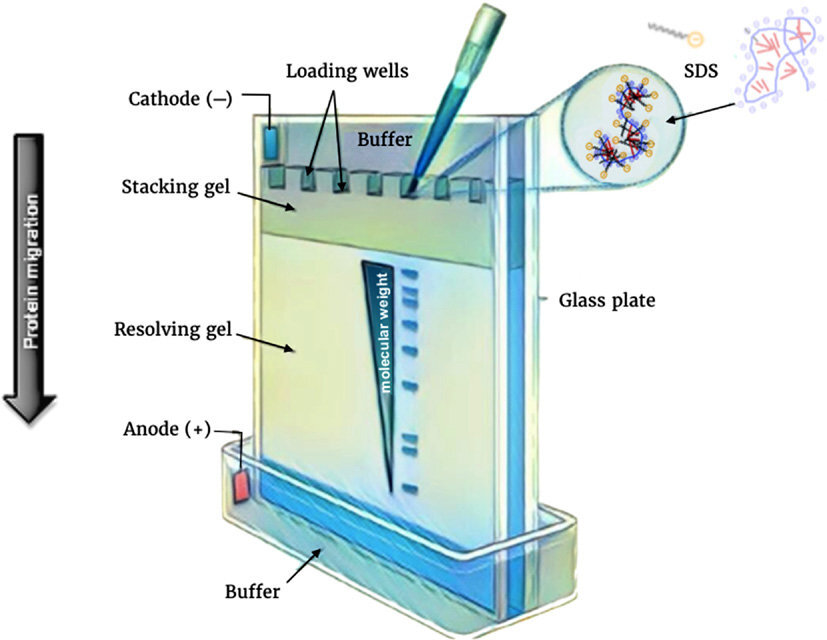
3. SDS-PAGE
SDS-PAGE (Sodium Dodecyl Sulfate Polyacrylamide Gel Electrophoresis) is a denaturing electrophoresis technique used to separate proteins according to their molecular weight.
SDS is a strong detergent that:
- unfolds proteins
- binds uniformly to polypeptide chains
- provides a constant negative charge
As a result, proteins migrate in the gel solely based on their size, not their natural charge.
Smaller proteins migrate faster through the gel matrix than larger ones.
After separation, proteins can be visualized using stains such as:
- Coomassie Brilliant Blue
- Silver staining
- Copper staining
Proteins can also be transferred onto membranes for Western blot analysis.
4. Isoelectric Focusing (IEF)
Isoelectric focusing separates proteins based on their isoelectric point (pI).
In this method, a pH gradient is established within a gel. When an electric field is applied:
- proteins migrate until they reach the region where pH = pI
- at that point, they stop moving because their net charge becomes zero
This technique provides extremely high resolution and can separate proteins differing by a single charge.
5. Two-Dimensional Gel Electrophoresis (2D Electrophoresis)
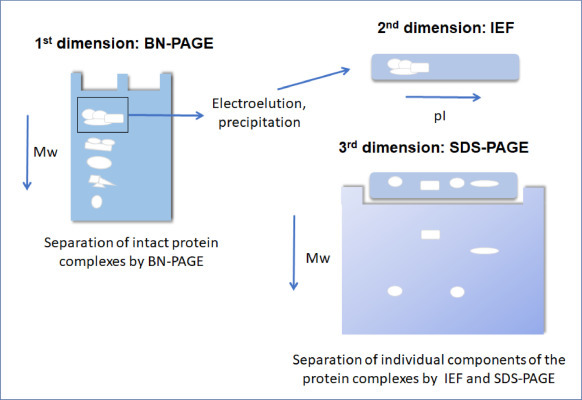 Two-dimensional electrophoresis combines two separation techniques:
Two-dimensional electrophoresis combines two separation techniques:
- Isoelectric focusing (separation by pI)
- SDS-PAGE (separation by molecular weight)
This powerful method allows researchers to resolve hundreds to thousands of proteins from complex biological samples.
It is widely used in:
- proteomics
- biomarker discovery
- disease research
6. Agarose Gel Electrophoresis
Agarose gel electrophoresis is mainly used for DNA and RNA analysis.
Agarose is a polysaccharide extracted from algae and forms a gel with relatively large pores.
This makes it ideal for separating large nucleic acid fragments ranging from approximately 0.2 kb to 50 kb.
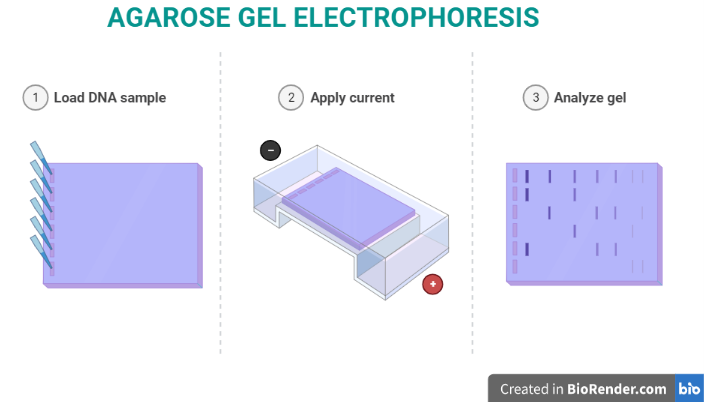
Typical applications include:
- PCR product analysis
- DNA fragment sizing
- restriction enzyme analysis
- DNA purification
DNA fragments are visualized by staining with fluorescent dyes such as ethidium bromide and observed under UV light.
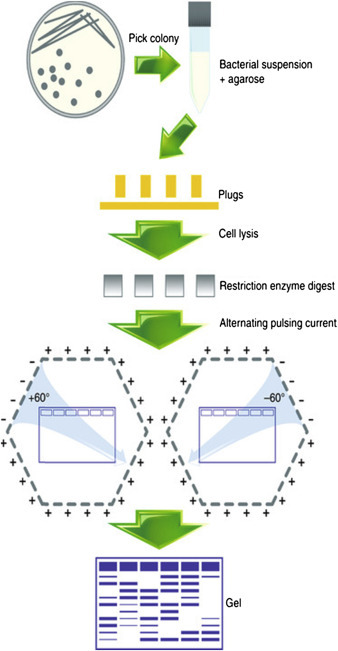
7. Pulsed-Field Gel Electrophoresis (PFGE)
Standard agarose gels cannot separate very large DNA molecules. Pulsed-field gel electrophoresis was developed to overcome this limitation.
In PFGE, the direction of the electric field is periodically changed. This allows extremely large DNA molecules (up to several megabases) to reorient and migrate through the gel.
PFGE is commonly used for:
- genome analysis
- microbial typing
- large DNA fragment separation
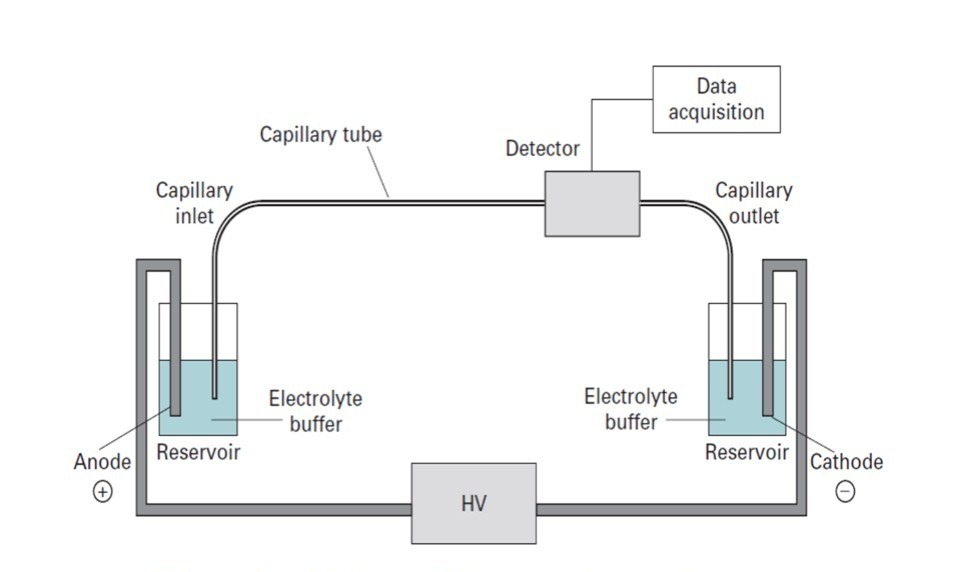
8. Capillary Electrophoresis
Capillary electrophoresis is a modern analytical technique that uses narrow capillary tubes filled with an electrolyte solution.
A high voltage is applied, allowing rapid separation of molecules based on their charge-to-size ratio.
Advantages include:
- very high resolution
- high sensitivity
- fast analysis times
- automation compatibility
Capillary electrophoresis is widely used in:
- pharmaceutical analysis
- clinical diagnostics
- genetic testing
- protein characterization
Applications of Electrophoresis in Biotechnology and Research
Electrophoresis techniques are fundamental tools in biological laboratories.
Common applications include:
- DNA analysis and sequencing
- protein purification and characterization
- clinical diagnostics
- genetic research
- enzyme analysis
- proteomics and biomarker discovery
These techniques allow scientists to visualize, quantify, and identify biomolecules with high precision.
Conclusion
Electrophoresis remains a cornerstone technique in molecular biology and biochemical research. By exploiting the movement of charged molecules in an electric field, scientists can separate complex mixtures of proteins and nucleic acids with remarkable accuracy.
From classical methods such as paper electrophoresis to advanced technologies like capillary electrophoresis and two-dimensional gels, these techniques continue to play a critical role in genomics, proteomics, biotechnology, and clinical diagnostics.
As analytical technologies evolve, electrophoresis will remain an essential tool for understanding the structure, function, and interactions of biological macromolecules.
Recent Posts
-
Histology Without Formalin
Histology Without Formalin: Challenges, Alternatives and Practical Considerations Introduction Tiss …25th Mar 2026 -
Ethidium Bromide as a Cooperative Effector of DNA Structure
Introduction DNA structure is not static. Under specific physicochemical conditions, DNA molecules c …25th Mar 2026 -
Electrophoresis Techniques in Molecular Biology
Electrophoresis is one of the most important analytical techniques used in molecular biology, bioche …25th Mar 2026