Human Platelet Lysate: A Safer and More Defined Alternative for Cell Culture
Human Platelet Lysate: A Safer and More Defined Alternative for Cell Culture
Introduction
In modern biomedical research, the shift toward safer, more ethical, and clinically compliant materials has become a defining priority. Traditional cell culture methods, once heavily reliant on animal-derived components, are now being reevaluated in light of regulatory pressures and scientific advancements. Among the emerging alternatives, Human Platelet Lysate (HPL) stands out as a transformative solution that bridges performance with safety. By harnessing the natural regenerative properties of human platelets, HPL offers researchers a biologically relevant and highly potent supplement that aligns with both experimental and therapeutic needs. Its growing adoption reflects a broader movement toward humanized systems in life sciences, where reproducibility, ethical sourcing, and translational potential are paramount.
What is Human Platelet Lysate?
Human Platelet Lysate is a complex, growth factor-rich supplement derived from human blood platelets, which are primarily known for their essential role in wound healing and tissue repair. Platelets contain intracellular granules packed with signaling molecules that regulate cell proliferation, migration, and differentiation. To produce HPL, platelet concentrates often obtained from blood donations are subjected to controlled lysis processes, such as repeated freeze-thaw cycles, which rupture the platelet membranes and release their bioactive contents. The resulting lysate is a concentrated mixture of growth factors like platelet-derived growth factor (PDGF), transforming growth factor-beta (TGF-β), vascular endothelial growth factor (VEGF), and epidermal growth factor (EGF), along with cytokines and adhesion molecules. This biochemical richness makes HPL a powerful supplement capable of mimicking the natural cellular microenvironment more closely than conventional sera.
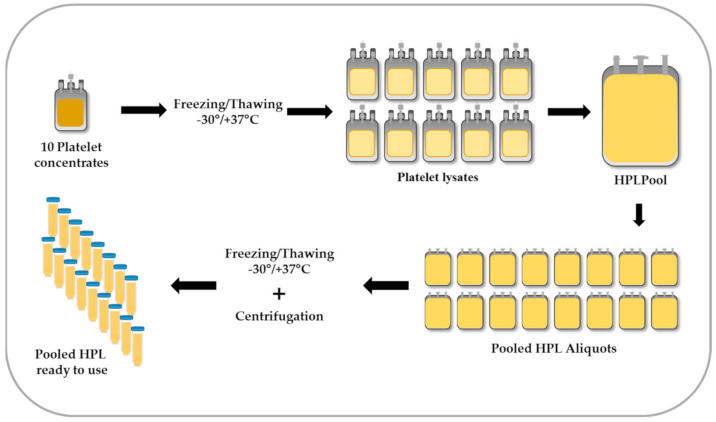
Figure : Preparation of pooled human platelet lysate (HPL): Fresh or expired platelet concentrates are subjected to freeze–thaw cycles to induce platelet lysis. Ten units of platelet lysate are then pooled into a single bag and aliquoted. Following a second freeze–thaw cycle, the bags are centrifuged to remove platelet debris. The final pooled HPL vials are stored at −30 °C or lower until use.
Why Use HPL Instead of Fetal Bovine Serum (FBS)?
For decades, Fetal Bovine Serum has been the cornerstone of in vitro cell culture due to its broad compatibility and nutrient content. However, its limitations have become increasingly difficult to ignore in a scientific landscape that prioritizes precision and ethics. HPL emerges as a compelling alternative by addressing these shortcomings on multiple fronts. Because it is derived from human sources, HPL provides a physiologically relevant environment for human cells, reducing species-specific discrepancies that can affect experimental outcomes. Moreover, HPL often supports faster and more robust cell proliferation, particularly in sensitive cell types like mesenchymal stem cells. From a safety perspective, it minimizes the risk of transmitting animal-borne pathogens and reduces immunogenic complications in clinical applications. These advantages position HPL not merely as a substitute, but as a superior evolution in cell culture supplementation.
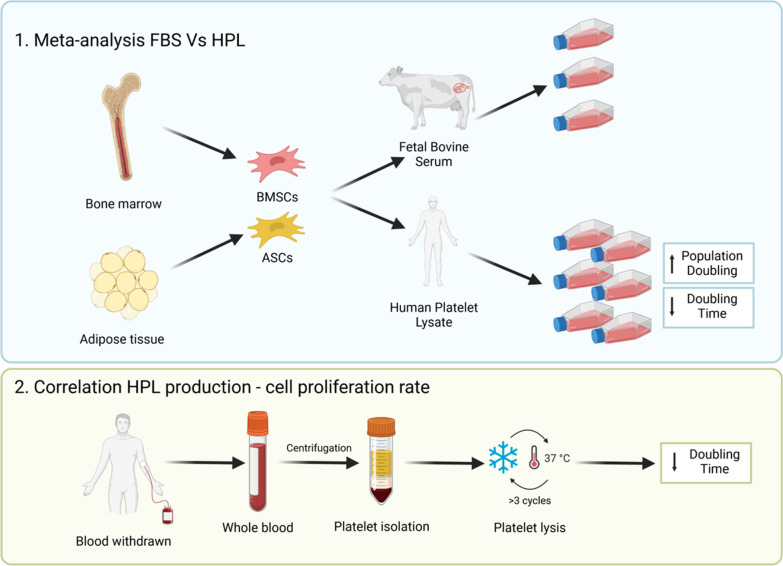
Figure : 1 The meta-analysis shows that HPL induces a population doubling increase and a doubling time decrease of both ASCs and BMSCs compared to FBS.
2. When at least 3 freeze/thaw cycles are applied to induce platelet lysis, the doubling time of HPL-cultured cells is lower than FBS-cultured cells .
Applications of Human Platelet Lysate
The versatility of Human Platelet Lysate has led to its widespread adoption across various domains of biotechnology and medicine. In stem cell research, HPL is particularly valued for its ability to enhance the expansion of mesenchymal stem cells while preserving their differentiation potential, a critical factor for regenerative therapies. In clinical and translational settings, it plays a key role in the development of cell-based therapies, where xeno-free conditions are essential for patient safety and regulatory approval. Beyond stem cells, HPL is also used in the culture of primary human cells, including fibroblasts, endothelial cells, and immune cells, where it supports both viability and functionality. Additionally, in biopharmaceutical research, HPL contributes to more predictive in vitro models, improving the reliability of drug testing and development pipelines. Its adaptability makes it an indispensable tool in advancing both fundamental research and applied sciences.
Challenges and Considerations
Despite its numerous advantages, the use of Human Platelet Lysate is not without challenges, and careful consideration is required to fully leverage its potential. One of the primary concerns lies in donor variability, as differences in platelet composition between individuals can lead to inconsistencies in lysate quality. To mitigate this, many suppliers pool platelet donations, creating more standardized batches that enhance reproducibility. Another technical consideration is the need for anticoagulants such as heparin in culture media, as HPL can induce gel formation due to fibrinogen content. Additionally, while HPL aligns well with regulatory frameworks, its production must adhere to stringent quality control measures, including pathogen screening and inactivation, which can increase costs. These factors highlight the importance of selecting high-quality, well-characterized HPL products for both research and clinical use.
Future Perspectives
Looking ahead, Human Platelet Lysate is poised to play an increasingly central role in the future of cell culture and regenerative medicine. Ongoing innovations aim to further standardize HPL production, reduce variability, and enhance its scalability for industrial and clinical applications. Advances in pathogen inactivation technologies and donor screening protocols are improving safety profiles, while efforts to develop chemically defined or semi-defined HPL formulations are pushing the boundaries of reproducibility. As the demand for xeno-free, GMP-compliant systems continues to rise, HPL is likely to become a cornerstone in next-generation bioprocessing and therapeutic development. Its integration into advanced manufacturing pipelines underscores its potential not only as a research tool but as a critical component in the delivery of cutting-edge medical treatments.
Conclusion
Human Platelet Lysate represents a significant advancement in the evolution of cell culture technology, offering a solution that aligns scientific performance with ethical responsibility. By providing a human-derived, growth factor-rich environment, HPL enhances cell proliferation, supports clinical translation, and reduces the risks associated with animal-derived supplements. While challenges related to standardization and cost remain, continuous improvements in production and quality control are steadily addressing these limitations. As the life sciences industry moves toward more human-relevant and regulatory-compliant practices, HPL stands out as a key enabler of innovation, driving progress in regenerative medicine, cell therapy, and beyond.
Recent Posts
-
HPLC Chromatography: Principle, Types, and Applications
HPLC Chromatography: Principle, Types, and Applications Introduction to HPLC Chromatography HPLC Chr …8th Apr 2026 -
Advances in Peptide Synthesis Technologies and Their Applications in Modern Biotechnology
Advances in Peptide Synthesis Technologies and Their Applications in Modern Biotechnology …27th Mar 2026 -
Western Blot
Introduction Western blotting is a fundamental analytical technique widely used in molecular biology …27th Mar 2026