Human Platelet Lysate in Regenerative Medicine: Applications and Benefits
Human Platelet Lysate in Regenerative Medicine: Applications and Benefits
Introduction
Regenerative medicine has emerged as one of the most exciting frontiers in modern healthcare, offering the potential to repair, replace, or regenerate damaged tissues and organs. Central to the success of these therapies is the ability to culture human cells effectively and safely. Traditionally, cell culture relied heavily on animal-derived supplements such as Fetal Bovine Serum (FBS). However, concerns about ethical sourcing, immune reactions, and clinical safety have prompted a search for human-derived alternatives. Human Platelet Lysate (HPL) has rapidly become a preferred solution. Rich in growth factors and cytokines, HPL provides a biologically relevant environment that supports cell proliferation, differentiation, and therapeutic potential, making it a cornerstone in regenerative medicine research and clinical applications.
What Makes HPL Ideal for Regenerative Medicine
Human Platelet Lysate is derived from human blood platelets, which naturally contain a reservoir of growth factors crucial for tissue repair. When platelets are lysed, they release molecules such as Platelet-Derived Growth Factor (PDGF), Transforming Growth Factor-beta (TGF-β), Vascular Endothelial Growth Factor (VEGF), and Epidermal Growth Factor (EGF). These factors play pivotal roles in stimulating cell proliferation, migration, and differentiation processes essential for tissue regeneration. Unlike animal sera, HPL is fully human in origin, reducing the risk of immune reactions and making it safer for clinical applications. Its composition also closely mimics the natural microenvironment of human tissues, enhancing the quality and functionality of cells cultured for therapeutic purposes.
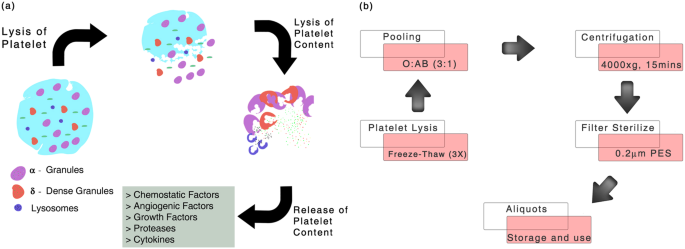
Figure: Platelet content and preparation of pooled human platelet lysate (pHPL). (a) Illustration showing the release of platelet granule contents which include cytokines, angiogenic factors, proteases and chemostatic factors on lysis (b) Overview of freeze-thaw mechanism of platelet lysate preparation, pooling and long-term storage.
Recent studies further support these advantages. For example, the article Human Platelet Lysate Supports Efficient Expansion and Stability of Wharton’s Jelly Mesenchymal Stromal Cells via Active Uptake and Release of Soluble Regenerative Factors demonstrates that HPL not only promotes efficient proliferation of Wharton’s Jelly-derived mesenchymal stromal cells (MSCs) but also maintains their phenotypic stability and functional properties. The study highlights that MSCs actively take up and release soluble regenerative factors from HPL, which reinforces their regenerative capacity and underscores the translational potential of HPL in clinical cell therapy and regenerative medicine applications.
Applications in Tissue Engineering and Stem Cell Therapies
One of the most prominent uses of HPL in regenerative medicine is in stem cell expansion, particularly mesenchymal stem cells (MSCs). MSCs are widely studied for their ability to differentiate into bone, cartilage, fat, and other tissues. HPL not only accelerates their growth compared to traditional FBS-supplemented media but also helps maintain their differentiation potential. This makes HPL an invaluable tool in tissue engineering, where generating functional, patient-ready tissues requires both quantity and quality. Additionally, HPL is used in organoid culture and other advanced 3D cell models, providing the signaling environment necessary for cells to self-organize and mimic human tissue architecture.
Benefits for Clinical Applications
HPL offers several advantages that directly impact clinical outcomes. Being xeno-free and human-derived, it minimizes the risk of transmitting animal-borne pathogens and reduces immunogenic responses when cells are transplanted into patients. Its rich growth factor content supports faster proliferation of therapeutic cells, which can shorten the time required to prepare cell therapies. Moreover, HPL can be produced in GMP-compliant conditions, ensuring consistency, safety, and regulatory readiness for clinical-grade applications. For hospitals and biotech companies, this means HPL not only improves cell culture efficiency but also facilitates compliance with stringent healthcare standards.
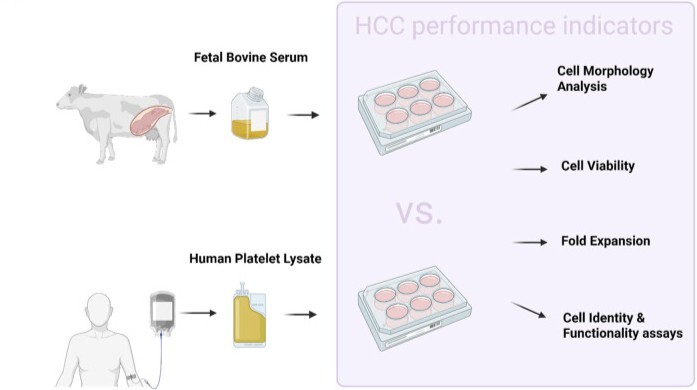
Figure: Data on human platelet lysate production methodology and human hematopoietic cell culture (HCC) conditions and performance indicators
Enhancing Wound Healing and Tissue Repair
Beyond laboratory cell culture, HPL has demonstrated direct benefits in wound healing and regenerative therapies. Its growth factors promote angiogenesis the formation of new blood vessels accelerating tissue repair in chronic wounds, burns, and surgical sites. HPL-based formulations are being explored for topical applications, injectable therapies, and scaffold-based tissue regeneration, showcasing its versatility. By harnessing the body’s natural healing molecules, HPL bridges the gap between laboratory cell expansion and practical clinical interventions, making regenerative treatments more effective and accessible.
Future Perspectives
The potential of Human Platelet Lysate in regenerative medicine continues to expand. Ongoing research focuses on standardizing production, improving batch consistency, and integrating HPL into automated cell culture systems. Innovations such as chemically defined HPL formulations and large-scale bioreactor production aim to make this human-derived supplement even more reliable and widely available. As regenerative therapies grow in scope and complexity, HPL is poised to play a pivotal role, supporting not only laboratory research but also the translation of therapies from bench to bedside.
Conclusion
Human Platelet Lysate has transformed the landscape of regenerative medicine by providing a human-derived, growth factor-rich supplement that enhances cell proliferation, differentiation, and tissue repair. Its applications span stem cell expansion, tissue engineering, organoid culture, and clinical therapies, all while offering a safer, xeno-free alternative to animal sera. By bridging laboratory innovation with patient-centered therapies, HPL is shaping the future of regenerative medicine and solidifying its role as a key enabler of safe, effective, and ethical biomedical solutions.
Recent Posts
-
HPLC Chromatography: Principle, Types, and Applications
HPLC Chromatography: Principle, Types, and Applications Introduction to HPLC Chromatography HPLC Chr …8th Apr 2026 -
Advances in Peptide Synthesis Technologies and Their Applications in Modern Biotechnology
Advances in Peptide Synthesis Technologies and Their Applications in Modern Biotechnology …27th Mar 2026 -
Western Blot
Introduction Western blotting is a fundamental analytical technique widely used in molecular biology …27th Mar 2026