Personalized Medicine Using Stem Cells and Engineered Tissues: Tailoring Treatments to Patients
Personalized Medicine Using Stem Cells and Engineered Tissues: Tailoring Treatments to Patients
Introduction
Personalized medicine represents a profound shift in the paradigm of healthcare, moving from generic treatment protocols toward patient-specific therapeutic strategies informed by an individual’s genetic, epigenetic, and cellular profile. In recent years, stem cell biology and tissue engineering have emerged as essential components of this movement, offering the potential to repair or replace damaged tissues in ways that are directly tailored to each patient. The convergence of these technologies allows clinicians and researchers to generate therapeutic constructs that mimic the patient’s own biology, reducing the risk of immune rejection and improving clinical outcomes. Unlike conventional therapies, which often treat symptoms rather than the underlying cause, patient-specific regenerative medicine provides a more precise approach by addressing the cellular and molecular deficits that contribute to disease. By integrating stem cell technology, engineered tissue scaffolds, and growth factor-mediated signaling, researchers are now capable of producing therapies that align with the patient’s intrinsic regenerative potential, thereby enhancing both safety and efficacy.
Introduction video :
Patient-Derived Stem Cells: The Foundation of Personalization
At the core of personalized regenerative medicine are patient-derived stem cells, including mesenchymal stem cells (MSCs), hematopoietic stem cells (HSCs), and induced pluripotent stem cells (iPSCs). iPSCs, for example, are generated by reprogramming somatic cells such as skin fibroblasts or peripheral blood mononuclear cells back to a pluripotent state through the introduction of key transcription factors (OCT4, SOX2, KLF4, and c-MYC). These cells retain the genetic signature of the patient, making them an ideal platform for autologous therapies. MSCs, often harvested from bone marrow, adipose tissue, or umbilical cord tissue, exhibit immunomodulatory properties in addition to multilineage differentiation potential, enabling applications ranging from cartilage repair to immune system regulation. The use of autologous stem cells reduces the likelihood of graft-versus-host reactions, alloimmune rejection, and the need for long-term immunosuppression. Furthermore, these cells can be genetically or epigenetically modified ex vivo to correct disease-causing mutations, enhance regenerative capacity, or overexpress therapeutic proteins, providing a highly customizable therapeutic platform.
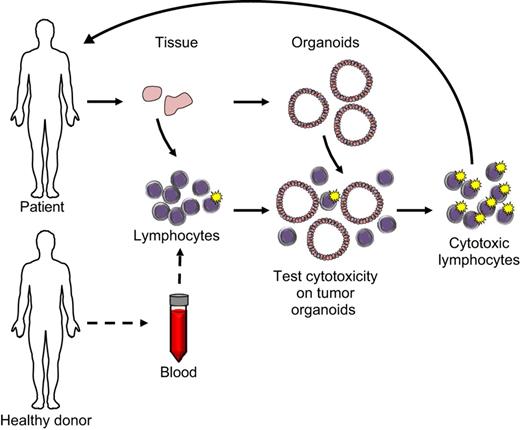
Figure: Exploiting adult stem cell-derived organoids for immunotherapy
Engineered Tissues: Customized Solutions for Repair and Replacement
Tissue engineering complements patient-derived stem cells by providing biomimetic scaffolds that support cell adhesion, proliferation, and differentiation. These scaffolds may be constructed from natural polymers like collagen, gelatin, or hyaluronic acid, synthetic biodegradable polymers such as polylactic-co-glycolic acid (PLGA), or hybrid materials that combine the mechanical strength of synthetics with the biological functionality of natural matrices. Cells seeded onto these scaffolds are exposed to biochemical cues, mechanical stress, and controlled microenvironments that guide tissue formation, differentiation, and maturation. In personalized therapies, scaffolds can be designed using patient imaging data (e.g., MRI, CT scans) to match the anatomical and biomechanical characteristics of the damaged tissue, enabling more precise integration upon transplantation. For example, cartilage repair constructs can be engineered to match the curvature, thickness, and zonal organization of a patient’s joint surface, while vascularized tissue constructs incorporate channels that support angiogenesis and nutrient delivery, improving graft survival and function. By combining cellular and structural customization, tissue engineering allows clinicians to move beyond symptomatic treatment to functional tissue replacement, restoring both anatomy and physiology.
Advancing Disease Modeling and Drug Testing
Personalized regenerative medicine extends beyond therapeutic interventions to patient-specific disease modeling. By differentiating iPSCs into organ-specific cell types or forming organoids, researchers can recapitulate the pathophysiology of diseases at the cellular and tissue levels. These patient-derived models provide a platform for high-throughput drug screening, toxicity testing, and predictive pharmacology, enabling clinicians to evaluate the efficacy of therapies before administration. For instance, in cardiology, iPSC-derived cardiomyocytes can reveal patient-specific drug responses to chemotherapeutic agents that may induce cardiotoxicity, while liver organoids can model metabolic diseases to predict hepatic drug metabolism and toxicity. Similarly, cancer organoids generated from a patient’s tumor cells allow for screening of chemotherapeutic or targeted therapies, facilitating precision oncology approaches that maximize therapeutic benefit and minimize side effects. By integrating stem cell-derived models with CRISPR-mediated gene editing, researchers can also explore disease mechanisms, validate potential drug targets, and correct genetic mutations in a controlled environment, bridging laboratory research and clinical application in a truly personalized manner.
Challenges and Considerations
While personalized regenerative medicine offers unprecedented opportunities, it faces several technical, regulatory, and logistical challenges. The generation of patient-specific stem cells and engineered tissues is resource-intensive, requiring sterile GMP-compliant facilities, specialized equipment, and skilled personnel. Standardizing protocols to ensure reproducibility and quality control across batches remains a major hurdle, particularly for autologous therapies where variability is inherent. Safety concerns include the potential for genomic instability, unintended differentiation, tumorigenicity, or immunogenic responses after transplantation. Regulatory frameworks are evolving to address these challenges, emphasizing rigorous preclinical testing, detailed characterization of cellular products, and validation of manufacturing processes. Additionally, the cost of personalized therapies can be prohibitive, although advances in automation, bioreactor expansion, 3D bioprinting, and off-the-shelf stem cell platforms are gradually improving scalability and accessibility.
Future Perspectives
The future of personalized regenerative medicine lies in the synergistic integration of stem cell technology, gene editing, tissue engineering, and computational modeling. Emerging techniques such as 3D bioprinting of vascularized tissues, CRISPR-based correction of patient-specific mutations, and organ-on-chip platforms are poised to expand the capabilities of personalized therapies. Advances in biomaterials and scaffold design will enable more precise replication of tissue microenvironments, while high-throughput screening and AI-driven modeling can optimize treatment strategies for individual patients. Ultimately, personalized regenerative medicine promises to deliver therapies that are predictive, curative, and precisely tailored to each patient’s biological context, revolutionizing treatment for genetic disorders, degenerative diseases, and traumatic injuries.
Conclusion
Personalized medicine using stem cells and engineered tissues is redefining therapeutic strategies by providing patient-specific, biologically relevant interventions. By combining autologous stem cells, customizable scaffolds, and advanced bioengineering techniques, researchers and clinicians can create therapies that are safer, more effective, and directly tailored to individual patients. Beyond treatment, these technologies enable disease modeling, predictive drug testing, and gene correction, positioning personalized regenerative medicine at the forefront of 21st-century healthcare. As innovations continue, the integration of biology, engineering, and precision medicine will unlock the full potential of patient-centered regenerative therapies, offering hope for conditions once deemed untreatable.
Recent Posts
-
Advances in Peptide Synthesis Technologies and Their Applications in Modern Biotechnology
Advances in Peptide Synthesis Technologies and Their Applications in Modern Biotechnology …27th Mar 2026 -
Western Blot
Introduction Western blotting is a fundamental analytical technique widely used in molecular biology …27th Mar 2026 -
Ethidium Bromide as a Cooperative Effector of DNA Structure
Introduction DNA structure is not static. Under specific physicochemical conditions, DNA molecules c …25th Mar 2026