Exosomes and Secretome in Tissue Regeneration: Cell-Free Therapeutic Strategies
Exosomes and Secretome in Tissue Regeneration: Cell-Free Therapeutic Strategies
Introduction
Regenerative medicine is undergoing a transformative shift, moving beyond traditional cell-based therapies toward cell-free therapeutic approaches that harness the bioactive potential of cellular secretions. Among these, exosomes and the broader secretome have emerged as powerful mediators of tissue repair and regeneration. Rather than transplanting whole cells, researchers are increasingly focusing on the molecules cells release proteins, lipids, nucleic acids, and extracellular vesicles that orchestrate healing processes at the molecular level. This approach offers a promising alternative that retains therapeutic efficacy while reducing many of the risks associated with live cell transplantation.
Understanding the Secretome: The Language of Cells
The secretome refers to the complete set of factors secreted by cells into their surrounding environment. This includes cytokines, growth factors, chemokines, extracellular matrix components, and extracellular vesicles such as exosomes. In regenerative contexts, stem cells particularly mesenchymal stem cells (MSCs) are known to exert much of their therapeutic effect not through direct differentiation, but through paracrine signaling. These secreted factors influence neighboring cells, modulate inflammation, stimulate angiogenesis, and promote tissue remodeling. Essentially, the secretome acts as a biochemical “communication system,” directing complex regenerative processes without requiring direct cell integration.
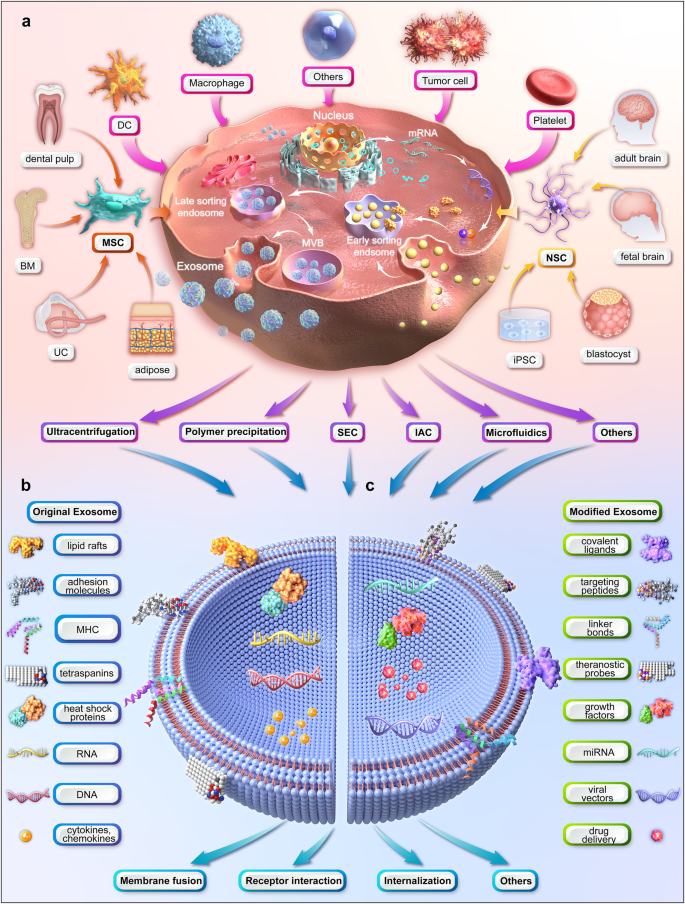
Figure: Illustration of the upstream measures of exosome therapy (figure generated using Autodesk 3ds Max 2023).
a production and purification of exosomes (MSCs and NSCs are used as examples for multipotent stem cells).
b content of natural exosomes.
c modification of exosomes. (BM bone marrow, DC dendritic cell, IAC immunoaffinity chromatography, iPSC induced pluripotent stem cell, MHC major histocompatibility complex, miRNA microRNA, MSC mesenchymal stem cell, MVB multivesicular body, NSC neural stem cell, SEC size-exclusion chromatography, UC umbilical cord)
Exosomes: Nano-Messengers of Regeneration
Within the secretome, exosomes play a particularly critical role. These nanoscale extracellular vesicles, typically 30–150 nm in diameter, are released by most cell types and carry a rich cargo of proteins, mRNA, microRNA, and lipids. Exosomes function as targeted delivery systems, transferring their molecular content to recipient cells and thereby altering gene expression and cellular behavior. In tissue regeneration, exosomes derived from stem cells have been shown to promote cell proliferation, angiogenesis, and immune modulation, making them highly attractive as therapeutic agents. Their small size and natural origin also allow them to penetrate tissues more effectively than whole cells.
Applications in Tissue Regeneration
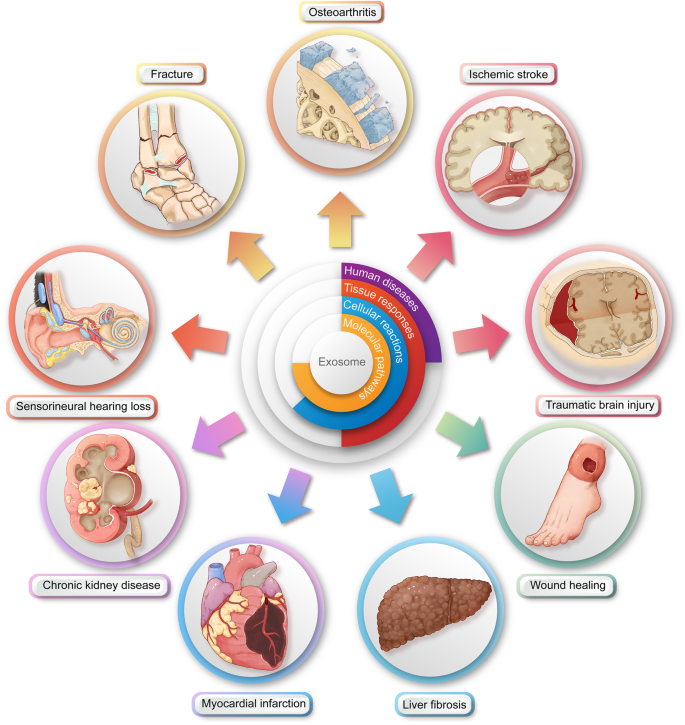
Exosome- and secretome-based therapies are being explored across a wide range of regenerative medicine applications. In cardiac repair, they help reduce fibrosis and enhance vascularization following myocardial infarction. In skin and wound healing, they accelerate tissue closure, stimulate collagen production, and reduce scarring. In musculoskeletal regeneration, including cartilage and bone repair, they support cell differentiation and matrix formation. Additionally, in neurological disorders, exosomes can cross the blood-brain barrier, offering a unique advantage for delivering therapeutic molecules to the central nervous system. These diverse applications highlight the versatility and potential of cell-free regenerative strategies.
Figure: Illustration of the downstream surgical applications of exosome therapy (figure generated using Adobe Photoshop 2023 and Adobe Illustrator 2023).
Advantages Over Cell-Based Therapies
One of the most compelling aspects of exosome and secretome therapies is their safety and scalability. Unlike living cells, exosomes are less likely to trigger immune rejection or form tumors, making them safer for clinical use. They can be stored, standardized, and manufactured at scale, addressing one of the major limitations of traditional cell therapies. Furthermore, their composition can be engineered or optimized to enhance specific therapeutic outcomes, offering a level of control that is difficult to achieve with whole-cell approaches. This positions exosomes as a next-generation platform in regenerative medicine.
Challenges and Current Limitations
Despite their promise, several challenges must be addressed before exosome-based therapies become widely adopted. Standardizing isolation, purification, and characterization methods remains a significant hurdle, as variations can impact therapeutic efficacy. Determining optimal dosing, delivery routes, and long-term safety profiles is also critical. Additionally, regulatory frameworks for cell-free biologics are still evolving, which can slow clinical translation. Researchers are actively working to overcome these challenges through improved bioprocessing technologies and rigorous clinical studies.
Future Perspectives
The future of regenerative medicine is likely to be heavily influenced by cell-free therapeutic strategies. Advances in bioengineering are enabling the design of engineered exosomes with enhanced targeting capabilities and customized cargo. Integration with technologies such as 3D bioprinting, biomaterials, and gene editing could further expand their therapeutic potential. As our understanding of intercellular communication deepens, exosomes and the secretome may become central tools in developing precision regenerative therapies tailored to individual patients.
Conclusion
Exosomes and the secretome represent a paradigm shift in regenerative medicine, offering a powerful, cell-free alternative to traditional therapies. By leveraging the natural communication systems of cells, these approaches can promote tissue repair, modulate immune responses, and enhance healing in a wide range of conditions. While challenges remain, ongoing research and technological innovation are rapidly advancing the field, bringing us closer to a future where regeneration is driven not by cells themselves, but by the signals they send.
Recent Posts
-
Advances in Peptide Synthesis Technologies and Their Applications in Modern Biotechnology
Advances in Peptide Synthesis Technologies and Their Applications in Modern Biotechnology …27th Mar 2026 -
Western Blot
Introduction Western blotting is a fundamental analytical technique widely used in molecular biology …27th Mar 2026 -
Ethidium Bromide as a Cooperative Effector of DNA Structure
Introduction DNA structure is not static. Under specific physicochemical conditions, DNA molecules c …25th Mar 2026